Amid an ongoing pandemic that has made headlines worldwide, there is a cause for celebration—over 1,000 patients admitted to a Carle Health facility with COVID-19 have been treated and discharged to recover at home.
For these patients and their families, this is a monumental achievement.
As the United States passes the one year mark since the first documented case of COVID-19 within its borders, researchers, physicians, and clinical experts around the country have made extraordinary advancements in how experts treat the virus. The healthcare community has learned, and is continuing to study, various new methods of approaching treatment and keeping COVID-19 positive patients healthy.
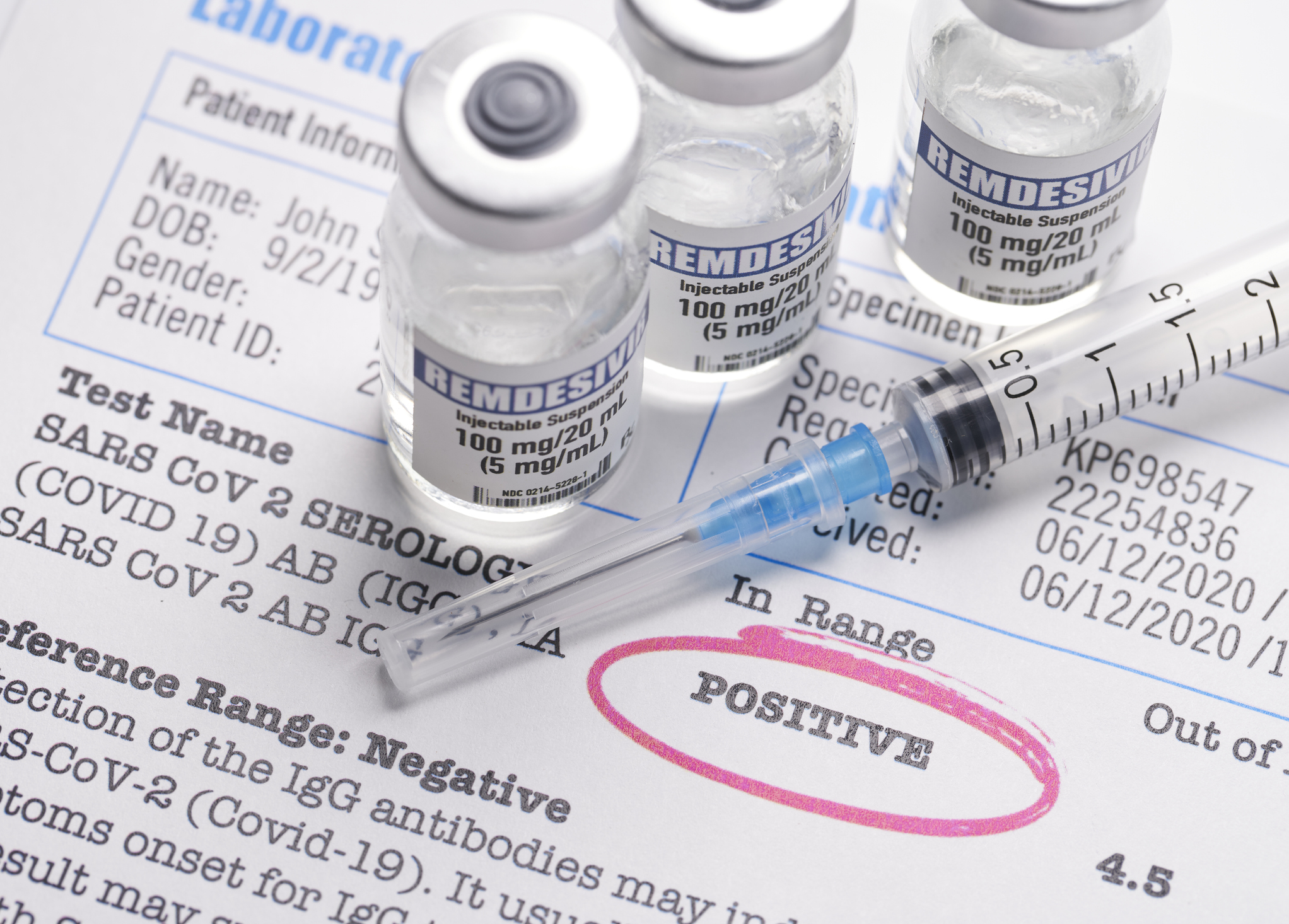
Carle is at the forefront of treatments and medications used to fight the virus, including two new treatments: Bamlanivimab and Remdesivir.

Robert Healy, MD, chief medical quality officer, Carle Health, says that while patients must fit very specific criteria for these treatments, those who do should take advantage of the opportunity.
“Since the beginning of the pandemic, healthcare professionals and researchers have constantly been learning and evaluating new ways to fight the virus and keep patients out of the hospital,” Healy said. “These three treatments have emerged as potentially lifesaving treatments against COVID-19, and we are pleased to be able to offer them right here at Carle.”
For many fighting the virus from home, Bamlanivimab, or commonly referred to as simply “Bam”, can be an easy way to fend off the virus’s symptoms with a prescribed medication. In short, Bamlanivimab works to block viral attachment and entry into human cells, therefore neutralizing the virus.
The Food and Drug Administration (FDA) has given Emergency Use Authorization (EUA) for Bamlanivimab, a one-time intravenous (IV) medical treatment, to treat COVID-19 patients with mild to moderate symptoms and a risk of severe complications.
Patients 12 and up weighing more than 88 lbs. and who are at high risk for progressing to severe COVID-19 and/or hospitalization are eligible for this treatment.
Bamlanivimab contains man-made antibodies similar to the antibodies of patients who recovered from COVID-19. Many scientists think that these antibodies may help limit the amount of virus in your body. This could give your body more time to learn how to make its own antibodies. It does not contain a live COVID-19 virus.
Patients are encouraged to contact their primary care provider to discuss if this treatment is right for them. Bamlanivimab is available to patients at Carle locations in Normal, Hoopeston, Urbana and Olney.
“I encourage those who have recently been diagnosed with COVID-19 to consider speaking with their provider about this option,” Healy said. “This treatment could significantly reduce the risk of suffering more severe complications that can lead to hospitalization.”
For those hospitalized with COVID-19, Remdesivir help speed up recovery and discharge. The drug is given through an IV to hospitalized patients with COVID-19. People typically receive an injection once a day for five to ten days, based on the severity of their condition.
The drug prevents the virus from producing a particular enzyme necessary for the virus to replicate itself. Once this happens, the virus is no longer able to spread within the body.
It’s important to consult with your healthcare provider if and when treatment is right for you. While these treatments may help those with COVID-19 recover, it’s important to remember to maintain proper safety measures to prevent the spread of the virus. Hand washing, practicing social distancing and wearing a mask is our best attack against COVID-19.
Visit carle.org/coronavirus for resources and more information on COVID-19.
For these patients and their families, this is a monumental achievement.
As the United States passes the one year mark since the first documented case of COVID-19 within its borders, researchers, physicians, and clinical experts around the country have made extraordinary advancements in how experts treat the virus. The healthcare community has learned, and is continuing to study, various new methods of approaching treatment and keeping COVID-19 positive patients healthy.
Carle is at the forefront of treatments and medications used to fight the virus, including two new treatments: Bamlanivimab and Remdesivir.

Robert Healy, MD, chief medical quality officer, Carle Health, says that while patients must fit very specific criteria for these treatments, those who do should take advantage of the opportunity.
“Since the beginning of the pandemic, healthcare professionals and researchers have constantly been learning and evaluating new ways to fight the virus and keep patients out of the hospital,” Healy said. “These three treatments have emerged as potentially lifesaving treatments against COVID-19, and we are pleased to be able to offer them right here at Carle.”
For many fighting the virus from home, Bamlanivimab, or commonly referred to as simply “Bam”, can be an easy way to fend off the virus’s symptoms with a prescribed medication. In short, Bamlanivimab works to block viral attachment and entry into human cells, therefore neutralizing the virus.
The Food and Drug Administration (FDA) has given Emergency Use Authorization (EUA) for Bamlanivimab, a one-time intravenous (IV) medical treatment, to treat COVID-19 patients with mild to moderate symptoms and a risk of severe complications.
Patients 12 and up weighing more than 88 lbs. and who are at high risk for progressing to severe COVID-19 and/or hospitalization are eligible for this treatment.
Bamlanivimab contains man-made antibodies similar to the antibodies of patients who recovered from COVID-19. Many scientists think that these antibodies may help limit the amount of virus in your body. This could give your body more time to learn how to make its own antibodies. It does not contain a live COVID-19 virus.
Patients are encouraged to contact their primary care provider to discuss if this treatment is right for them. Bamlanivimab is available to patients at Carle locations in Normal, Hoopeston, Urbana and Olney.
“I encourage those who have recently been diagnosed with COVID-19 to consider speaking with their provider about this option,” Healy said. “This treatment could significantly reduce the risk of suffering more severe complications that can lead to hospitalization.”
For those hospitalized with COVID-19, Remdesivir help speed up recovery and discharge. The drug is given through an IV to hospitalized patients with COVID-19. People typically receive an injection once a day for five to ten days, based on the severity of their condition.
The drug prevents the virus from producing a particular enzyme necessary for the virus to replicate itself. Once this happens, the virus is no longer able to spread within the body.
It’s important to consult with your healthcare provider if and when treatment is right for you. While these treatments may help those with COVID-19 recover, it’s important to remember to maintain proper safety measures to prevent the spread of the virus. Hand washing, practicing social distancing and wearing a mask is our best attack against COVID-19.
Visit carle.org/coronavirus for resources and more information on COVID-19.
Categories: Staying Healthy
Tags: Bamlanivimab, Bloomington-Normal, Champaign-Urbana, COVID-19, Hoopeston, Olney, Remdesivir, treatment